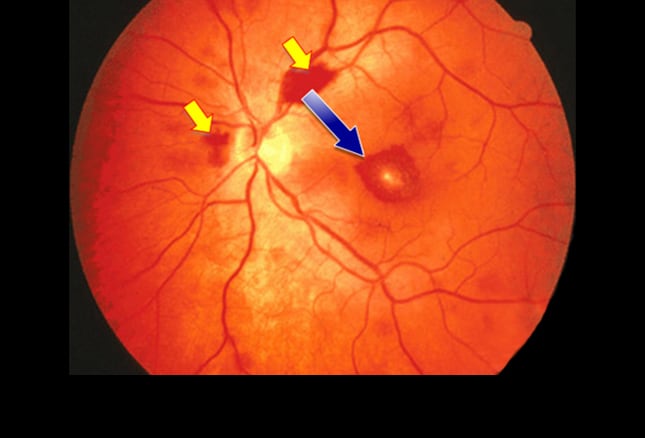
However, no information is available on associations between retinal hemorrhagic pattern and perfusion status a reason may be because it is difficult to evaluate hemorrhagic patterns objectively. Histologically, hemorrhages in the retinal nerve fiber layer are linear because the blood is aligned parallel to the axons, and rounded dot and blot hemorrhages lie in the nuclear and plexiform layers 19. A characteristic feature of the retinal hemorrhages is that they are often referred to as being flame-shaped (fan-shaped or splinter-shaped), and occasionally as a blot or dot (non-flame-shaped) 17, 18. The posterior poles of eyes with acute BRVO exhibit simultaneous retinal ischemia and retinal hemorrhages 17, 18. In addition, color fundus photography, the most common imaging modality, has not been applied to the analysis of NPAs. However, the interpretations and analysis of these images are often complicated, and parts of these modalities are commonly unavailable 13, 14, 15, 16. Some investigators reported that red-free imaging 12, or angiography 13 and en-face 14, 15, 16 optical coherence tomography (OCT) images were useful to detect NPAs in eyes with retinal vascular diseases. However, FA requires intravenous injections of dye, and is therefore too invasive to be performed readily 7, 8, 9, 10, 11 the development of alternative techniques is thus sought. In the management of BRVO, it is thus worthwhile to evaluate macular perfusion status in the acute phase.įluorescein angiography (FA) is still an essential tool to detect morphologic and functional changes of the retinal vasculature. Previously, Ota and associates reported that macular function measured by microperimetry was markedly decreased in macular nonperfused areas (NPAs) of eyes with BRVO 5, although Campochiaro and associates recently reported that anti-VEGF treatments could prevent NPA enlargement and promote reperfusion 6. However, despite the use of aggressive anti-VEGF treatments, we still encounter cases with inadequate recovery of visual functions in which retinal ischemia occasionally involves the macular area. The visual prognosis of branch retinal vein occlusion (BRVO) has been substantially improved since anti-vascular endothelial growth factor (VEGF) agents were introduced 1, 2, 3, 4. In conclusion, we objectively evaluated retinal hemorrhagic patterns at the posterior pole in BRVO using the parallelism method, which was useful in differentiating macular perfusion status. The area under the curve for parallelism was 0.975 ( P < 0.001), suggesting an accurate diagnostic parameter for macular perfusion differentiation. Parallelism in eyes with a flame-shaped hemorrhage was higher than in those with a non-flame-shaped hemorrhage ( P < 0.001), and in those with a nonischemic macula versus those with an ischemic macula ( P < 0.001). Of the 19 eyes classified as having a non-flame-shaped hemorrhage, 13 (68.4%) had an ischemic macula and 6 (31.6%) had a nonischemic macula ( P < 0.001). All 39 eyes with a flame-shaped hemorrhage showed a nonischemic macula. Predominant hemorrhagic patterns were flame-shaped in 39 (67.2%) and non-flame-shaped in 19 (32.8%) eyes. Macular perfusion was judged as nonischemic in 30, ischemic in 28, and undeterminable in 5 among the 63 eyes. 
Retinal hemorrhagic patterns and macular perfusion status were evaluated via fundus photography and fluorescein angiography, respectively. This prospective study included 63 eyes with acute branch retinal vein occlusion (BRVO) to evaluate the retinal hemorrhagic patterns at the posterior poles and explore their clinical relevance in macular perfusion differentiation.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
